Sleep looks evolutionarily absurd.
It imposes paralysis, blindness, and vulnerability. It consumes time that could be spent feeding, mating, escaping, competing. Yet it is nearly universal across nervous systems. Something deeper than “rest” must be at stake.
The SPIN framework proposes a stark answer:
Sleep preserves the operating regime of the brain.
During wakefulness, cortical population activity drifts: synapses are modified, excitation–inhibition balance shifts, covariance structure reorganizes, and task-relevant manifolds gradually rotate in state space. Within-session decoding may remain intact, yet cross-session alignment erodes and flexible cognition becomes fragile before stored content is lost. Wake is a drift-inducing regime.
Non-REM sleep—especially slow-wave sleep—introduces a global contraction dynamic. Slow oscillation down–up cycles create low-information epochs that renormalize population statistics, re-centering gain, shared variance, and alignment structure. But stabilization alone is not enough. Within this constrained regime, hippocampal sharp-wave ripples coordinated with thalamocortical spindles provide windows for selective plasticity, integrating behaviorally relevant ensembles without destabilizing global geometry. REM further supports cross-network reorganization and abstraction.
Sleep is therefore a control architecture: SWS re-centers population geometry, while ripple–spindle coupling and REM refine content within it. Without contraction, integration amplifies drift; without integration, contraction preserves a static scaffold. The brain does not sleep merely to rest—it sleeps to remain a portable, adaptable dynamical system.
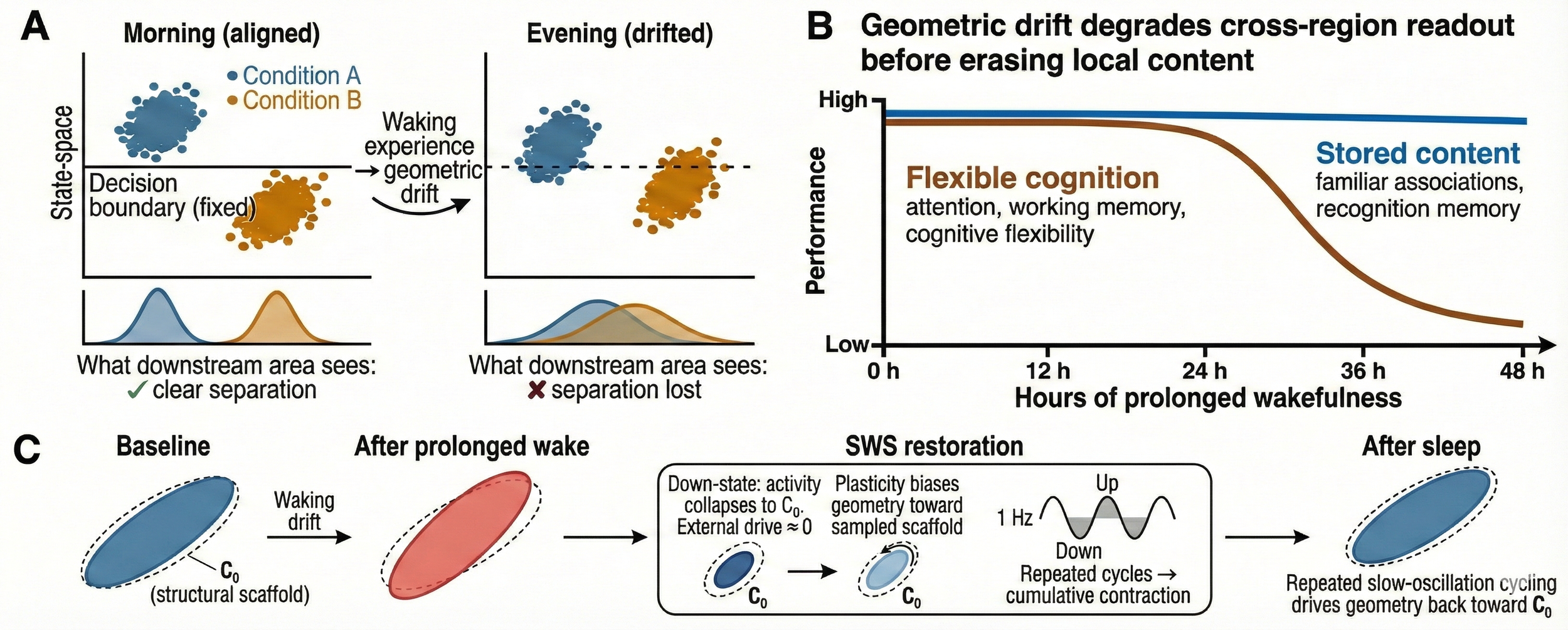

Figure 1 | Wake-induced geometric drift reduces population-geometry portability, and NREM/SWS restores alignment toward a structural scaffold. (a) Conceptual schematic of population activity in a low-dimensional “state-space” for two conditions (A, B). In the morning (aligned), a fixed downstream readout (decision boundary) cleanly separates conditions; the corresponding 1D projections (bottom) show minimal overlap. Across waking experience, representations can drift (e.g., rotate/shear the task-relevant subspace), such that within-session decodability may remain high but a largely fixed downstream readout becomes progressively mismatched, yielding lost separation in the evening (drifted). (b) Behavioral dissociation during prolonged wakefulness: performance in flexible cognition (attention, working memory, cognitive flexibility) declines earlier/steeper than performance supported by stored content (familiar associations, recognition memory). This is interpreted as geometric drift degrading cross-region readout before erasing locally decodable content. (c) Hypothesized mechanism for portability restoration. Baseline population geometry is biased toward a scaffold-preferred covariance/subspace, C₀ (“structural scaffold”). Wake-induced drift perturbs geometry away from this basin; repeated slow-oscillation down–up cycling during NREM slow-wave sleep (SWS) provides low-input epochs that preferentially sample scaffold-dominated dynamics and is hypothesized to drive a contraction back toward C₀, partially restoring cross-session decoder transfer and manifold alignment upon return to wakefulness. All panels are schematics. NREM, non–rapid eye movement; SWS, slow-wave sleep; C₀, scaffold-preferred covariance/subspace.